Lead Author
Institution
Published
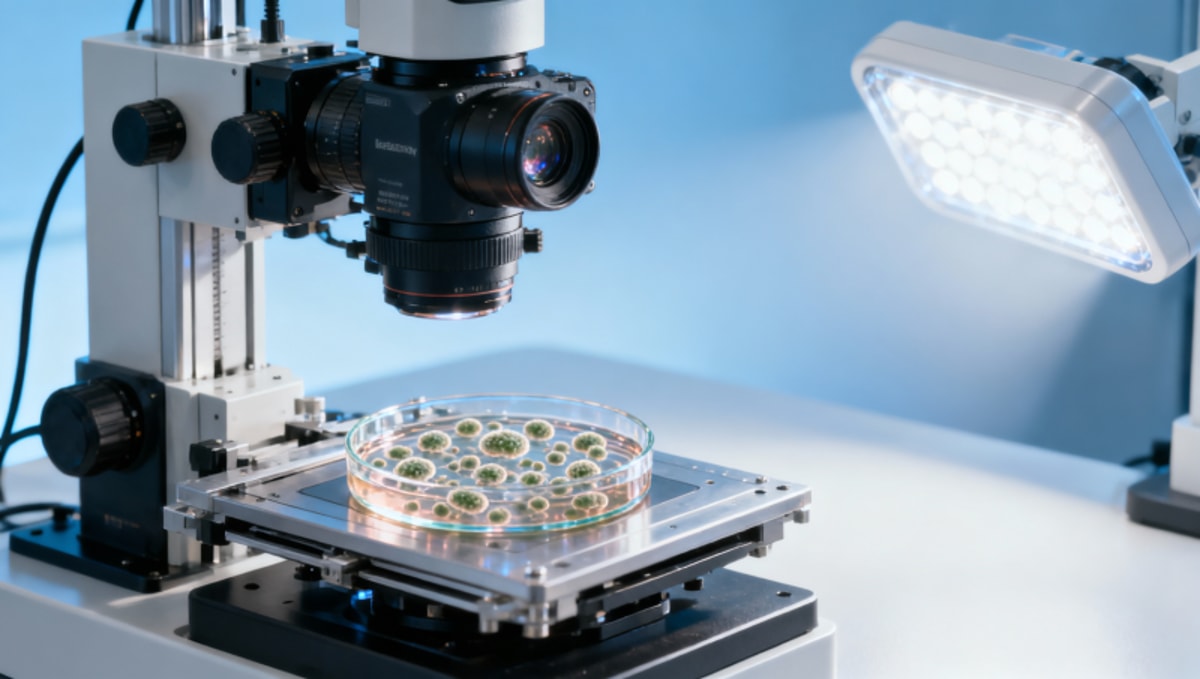
Abstract
Avoiding common errors in a high-speed camera cell tracking setup is essential for reliable imaging, data integrity, and downstream lab decisions. From motion blur and calibration drift to lighting mismatch and software sync issues, these mistakes can distort biological analysis and equipment evaluation. For researchers, operators, and procurement teams, understanding high-speed camera cell tracking pitfalls also supports broader performance benchmarking across modern laboratory systems.
Why do high-speed camera cell tracking setups fail in real laboratory workflows?
A high-speed camera cell tracking setup often looks correct on paper but underperforms during live acquisition. In bioscience labs, the issue is rarely a single defective part. More commonly, 3 to 5 small mismatches accumulate across optics, illumination, stage motion, trigger timing, and analysis software. When these mismatches occur together, the result is unreliable cell trajectory data, inconsistent frame-to-frame contrast, and poor reproducibility between operators or sites.
Operators usually notice the problem first as blurred edges, unstable tracking masks, or dropped frames during runs lasting 10 to 30 minutes. Technical evaluators may detect a different symptom: benchmark tests that looked acceptable at static resolution do not hold under dynamic imaging. Procurement teams then face a familiar challenge. They may compare camera specifications only by frame rate, yet cell tracking quality depends just as much on exposure control, pixel sensitivity, synchronization architecture, and calibration stability.
In medical technology and life science environments, setup errors also create a compliance and decision risk. If a laboratory uses image-derived cell motility data to support process development, assay validation, or equipment qualification, weak traceability can affect internal quality reviews and cross-department acceptance. This is why independent technical repositories such as G-MLS matter. Procurement directors, laboratory heads, and med-tech engineers need verifiable evaluation criteria, not only promotional vendor claims.
From a practical standpoint, most failures in a high-speed camera cell tracking setup fall into 4 categories: image formation errors, timing and synchronization errors, environmental instability, and software interpretation errors. Understanding these categories helps stakeholders define whether they need reconfiguration, retraining, component replacement, or a broader system-level review before the next procurement cycle.
The most common failure patterns seen across evaluation projects
- Exposure is too long for the target motion, causing motion blur even when nominal frame rate is high. A system running at 500 fps can still blur fast-moving cells if exposure remains poorly matched.
- Illumination intensity is uneven across the field of view, leading to biased segmentation and false velocity calculations in edge regions.
- Trigger timing between camera, stage, and external devices drifts over repeated sessions, especially after maintenance, software updates, or cable changes.
- Pixel size, magnification, and calibration scale are not revalidated after lens replacement, producing measurement errors that may remain hidden for weeks.
These errors are especially relevant in multi-user facilities, contract labs, translational research centers, and hospital-associated laboratories where one instrument may serve 2 to 4 different assay types. A setup that appears stable for suspension cells may not be adequate for adherent cell migration studies, microfluidic observation, or fast deformation events. The setup must therefore be assessed against application conditions, not generic performance sheets.
Which setup mistakes distort cell tracking data the most?
The most damaging mistakes are the ones that preserve image visibility but corrupt measurement integrity. In other words, the video still looks usable to the human eye, yet the extracted cell paths, speed distributions, or displacement curves are wrong. This is why high-speed camera cell tracking setup mistakes frequently pass unnoticed during short demos and only become evident during batch analysis, method transfer, or procurement acceptance testing.
The first high-impact mistake is inadequate control of exposure versus motion. A short exposure may require stronger illumination, but without that tradeoff the system records smeared cell boundaries. The second is calibration neglect. If the pixel-to-micron relationship is not confirmed after changing the objective, tube lens, or camera mount, quantitative outputs no longer represent real movement. The third is poor synchronization between camera capture and external events such as stimulation pulses, microfluidic flow changes, or stage repositioning.
The table below summarizes common setup mistakes in high-speed camera cell tracking, their technical consequence, and the operational impact on bioscience workflows. It is useful for information researchers, technical reviewers, and buyers who need a fast screening tool before deeper validation.
For procurement and quality teams, this table highlights an important point: many imaging failures are not camera-only failures. A high-speed camera cell tracking setup is a system purchase, not a single-component purchase. Evaluation should therefore cover at least 5 linked elements: sensor, optics, illumination, control electronics, and analysis environment. This system view is central to the benchmarking approach used by G-MLS across imaging and laboratory technologies.
What operators should check before blaming the camera
1. Optical path integrity
Verify objective cleanliness, tube alignment, and focus stability after warm-up. In many labs, 15 to 30 minutes of thermal stabilization is enough to reveal whether drift is mechanical or environmental. If the field stays sharp at the center but degrades near the edges, the root cause may be lens mismatch or mounting tension rather than sensor performance.
2. Illumination consistency
Check whether brightness remains stable over continuous runs, especially across 5 to 10 repeated captures. LED flicker, poor diffuser selection, or reflective vessel surfaces can alter apparent cell borders. A brighter system is not automatically a better system if the illumination profile is uneven.
3. Software and trigger alignment
Confirm that timestamps, stage commands, and image sequence metadata agree. Even a small synchronization gap can shift derived cell paths in dynamic assays. This matters when data will later support engineering qualification, method comparison, or procurement acceptance criteria.
How should technical evaluators and buyers compare setup quality before procurement?
When comparing systems, many buyers focus on headline specifications such as maximum frame rate, sensor megapixels, or software feature count. Those figures matter, but they do not tell the full story. For a high-speed camera cell tracking setup, a better procurement model uses 3 layers of assessment: imaging quality under motion, quantitative reliability across repeated runs, and implementation fit for the intended laboratory environment.
A procurement review should also distinguish between research flexibility and operational stability. A research team may accept manual adjustment if it gains experimental freedom. A hospital lab, GMP-adjacent facility, or centrally managed core lab often needs repeatability across multiple users, tighter documentation, and easier maintenance. That changes the evaluation matrix significantly.
The following table provides a practical selection framework. It compares what buyers often ask first with what they should verify before approving budget, installation, or vendor shortlisting for a high-speed camera cell tracking setup.
This selection approach reduces the risk of buying a technically impressive but operationally fragile system. It also supports cross-functional approval because the same matrix is useful for operators, engineers, project managers, and purchasing teams. G-MLS applies this type of structured assessment across advanced imaging and life science research tools, helping organizations compare equipment against standards, workflow fit, and long-term maintainability.
A practical 6-point procurement checklist
- Confirm the target assay type, expected cell speed, and recording duration before selecting camera frame rate and storage architecture.
- Require evidence of repeatability across at least 3 separate runs, not only a single demonstration video.
- Review whether the optical path can support your intended vessel types, microfluidic chips, or culture formats without repeated adapter changes.
- Check whether calibration and maintenance procedures are documented clearly enough for quality and safety teams.
- Assess data export compatibility with the laboratory’s existing analysis or archival environment.
- Clarify spare parts, service response windows, and post-install support for the first 2 to 4 quarters of operation.
For commercial evaluators and enterprise decision-makers, this checklist translates technical risk into procurement language. It helps estimate training burden, downtime exposure, validation effort, and upgrade viability. In many organizations, these factors influence return on investment more than the initial hardware price alone.
What implementation and compliance issues are often overlooked after installation?
Even a well-selected high-speed camera cell tracking setup can underperform after delivery if implementation is rushed. Typical post-install mistakes include skipping baseline acceptance tests, failing to document calibration status, and allowing users to change optics or software settings without version control. In regulated or quality-managed environments, these are not minor issues. They affect traceability, reproducibility, and internal confidence in image-derived results.
A robust implementation plan usually needs 4 stages: site readiness review, installation and alignment, operational qualification, and user training with controlled SOP release. Depending on complexity, this process may take 7 to 15 days for a straightforward laboratory deployment or 2 to 4 weeks when integration with external controllers, environmental chambers, or data systems is required. Compressing that timeline can create months of avoidable troubleshooting.
For medical and life science organizations, standard awareness is also important. While the exact regulatory pathway depends on use case, quality teams commonly reference general frameworks such as ISO 13485 for quality management relevance, FDA expectations for documented controls in applicable contexts, and CE MDR awareness where device-linked workflows are involved. The point is not to overstate regulatory scope, but to ensure the setup and its documentation are compatible with disciplined technical review.
This is one reason G-MLS emphasizes cross-sector data transparency. A laboratory camera setup does not exist in isolation. It interacts with sample handling, analytical interpretation, documentation practice, and procurement governance. By benchmarking hardware and workflows against recognized standards and engineering logic, teams can avoid fragmented decisions that create hidden operational cost later.
Implementation controls that reduce long-term error risk
Document 6 acceptance items
At minimum, verify image clarity under motion, scale calibration, trigger response, illumination uniformity, file integrity, and operator handoff records. These 6 items create a practical baseline for future troubleshooting and supplier discussions.
Set review intervals
A quarterly review is often appropriate for active shared-use systems, while lower-use setups may be checked every 6 months. Review intervals should tighten after software changes, lens replacement, service work, or relocation.
Control user changes
Limit unrestricted parameter edits. If multiple users can alter exposure, gain, trigger mode, and analysis thresholds without logging, data comparability declines quickly. Controlled profiles help maintain consistency across projects and departments.
FAQ: how to reduce high-speed camera cell tracking setup mistakes before they affect decisions
How do I know whether blur comes from focus problems or exposure problems?
Check static targets first, then dynamic samples. If static sharpness is acceptable but moving cells remain smeared, exposure is the more likely issue. If both static and dynamic images degrade, inspect focus, alignment, vibration, and thermal drift. Running 3 short tests with different exposure settings often reveals the pattern faster than changing lenses immediately.
What frame rate is enough for a high-speed camera cell tracking setup?
There is no universal single number because required frame rate depends on cell behavior, magnification, and the event being measured. Buyers should ask for a usable operating window, not just a peak fps value. A system that performs reliably at the needed resolution and exposure range is more valuable than one advertising a much higher frame rate under impractical conditions.
What should procurement teams request from vendors or evaluators?
Request 5 things: application-matched demo conditions, calibration method description, synchronization details, expected support in the first 30 to 90 days, and a clear statement of required accessories or environmental conditions. Without these details, price comparisons are incomplete and post-install surprises become more likely.
Can software alone fix poor cell tracking images?
Software can improve segmentation and reduce analysis burden, but it cannot reliably restore information never captured by the optical system. If blur, glare, or timing error is severe, downstream algorithms may only create cleaner-looking but still inaccurate outputs. Correct acquisition remains the foundation of valid measurement.
Why choose G-MLS when reviewing imaging risks, specifications, and procurement options?
G-MLS supports organizations that need more than vendor brochures and isolated product claims. As an independent technical repository and academic intelligence hub focused on medical technology and bioscience, G-MLS helps teams evaluate high-speed camera cell tracking setups in the broader context of laboratory performance, engineering integrity, and standards-aware decision-making. This is especially valuable when multiple departments must agree on one imaging investment.
For information researchers, G-MLS helps clarify which parameters actually affect image-based cell analysis. For operators and service teams, it supports practical review of failure points such as lighting mismatch, trigger instability, and calibration drift. For procurement, project, and commercial teams, it provides a structured framework for comparing systems across application fit, compliance readiness, maintenance burden, and total implementation risk.
You can contact G-MLS for targeted support on parameter confirmation, product selection logic, implementation planning, documentation expectations, delivery-cycle discussion, standards-oriented benchmarking, and quotation alignment for imaging-related laboratory systems. If your team is reviewing a new high-speed camera cell tracking setup or troubleshooting an existing one, a structured technical assessment can shorten decision time and reduce costly configuration errors.
If needed, prepare 4 basic inputs before reaching out: your cell tracking application, expected motion speed or event type, current optical configuration, and whether the system must meet internal quality or regulated documentation requirements. With those details, the discussion can move quickly from broad specification lists to practical selection, validation, and deployment guidance.
Recommended News
Metadata & Tools
Related Research
- Sample Preparation System OEM and After-Sales RiskSample preparation system OEM risk guide: compare after-sales support, automated pipetting CV, centrifuge rotor imbalance limits, and spectrophotometer wavelength accuracy before you buy.
- How to Compare Sample Preparation System OEM OffersSample preparation system OEM comparison guide: evaluate automated pipetting CV, spectrophotometer wavelength accuracy, compliance, service, and TCO to choose a reliable lab partner.
- Sample Preparation System OEM: Build or Outsource?Sample preparation system OEM: build or outsource? Compare cost, compliance, automated pipetting CV, spectrophotometer wavelength accuracy, and centrifuge rotor imbalance limits.
- Lab Labeling System Factory and Traceability NeedsLab labeling system factory selection shapes traceability, compliance, and workflow reliability. Learn how to assess integration, durability, support, and total lifecycle value.
- Should You Switch Lab Labeling System Factories?Lab labeling system factory switching guide: compare compliance, uptime, integration, and supply risk alongside sample preparation system OEM, digital pipette manufacturer, and vacuum manifold factory options.
